Avoiding endotoxin contamination in Health care and pharmaceutical options calls for subsequent sterilization tactics and environmental criteria.
Endotoxins are a significant virulence factor of gram-detrimental microbes, as their release may well result in a cascade of fever, inflammation, along with other immune responses from the host, possibly resulting in ailment. They are a significant mediator of sepsis and septic shock.
Examples contain antibiotics made by fermentation or perhaps the by-items of gram negative micro organism utilised to provide genetically engineered drug products and solutions. The likely use of yeast In this particular region is remaining evaluated to get rid of this issue.
Mild scattering gives distinct positive aspects for researchers serious about immunology. Learn the way the NEPHELOstar Moreover is utilized for high-throughput immunological tests.
endotoxin, harmful material bound to the bacterial mobile wall and unveiled once the bacterium ruptures or disintegrates. Endotoxins consist of lipopolysaccharide and lipoprotein complexes.
These methods are definitely the Gel-Clot method, the turbidimetric method as well as colorimetric method, which may be completed inside a qualitative or quantitative manner.
This method can be used in numerous types of matrices and has the advantage of with the ability to measure the kinetics from the reaction. Therefore, this method is a lot more specific compared to Gel Clot method. Utilizing the accesories and reagents marketed with the brand PYROSTAR™, we will evaluate numerous samples concurrently. The Toxinometer® ET-6000 Series makes it possible for us to read more read on multi-effectively plates in a very wide selection of endotoxin concentrations, managing the temperature at which the test is executed.
This human-primarily based technique is particularly valuable when evaluating the prospective scientific effect of endotoxin contamination.
For parenteral products and solutions, inspections have demonstrated that exactly where pyrogen complications were being present in dosage sorts, and once the resource was one of many Uncooked supplies, it absolutely was the active drug material. This was particularly real for drug substances in which approach h2o was employed at some late phase in the synthesis approach.
Launch criteria: For those who laboratory provides a release conditions metric, it really is recommended to include a sample at this focus in your common curve. This tends to assure you can have the very best volume of self-assurance that not known samples are above or beneath this threshold.
Good dilution: Dilute samples appropriately to prevent inhibition or improvement of your assay response. Validate dilution factors in the course of assay growth.
M signifies the rabbit pyrogen test dose or the maximum human dose per kilogram that might be administered in a single a person hour period, whichever is larger. If an item is labeled for intrathecal injection, then K is 0.2 EU/kg. Nevertheless, you will discover five water solutions (previously reviewed) which, because of the significant volumes that may be detection of bacterial endotoxins administered and absence of dose limitation, have precise endotoxin limits for each ml.
As with parenteral drug merchandise, sterile equipment have occasionally been shown to become contaminated with endotoxins. Sources have already been drinking water which in some way entered in the production course of action.
The results of endotoxin are related to the amount of endotoxin within the merchandise dose administered to your client. As the dose may differ from products to products, the endotoxin Restrict is expressed as K/M. K is five.0 EU/kilogram (kg.), which signifies the approximate threshold pyrogen dose for human beings and rabbits. That is the degree at which a product is adjudged pyrogenic or non-pyrogenic.
 Taran Noah Smith Then & Now!
Taran Noah Smith Then & Now!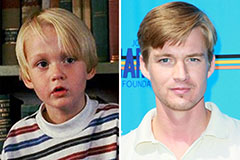 Mason Gamble Then & Now!
Mason Gamble Then & Now! Molly Ringwald Then & Now!
Molly Ringwald Then & Now!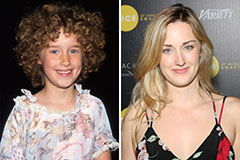 Ashley Johnson Then & Now!
Ashley Johnson Then & Now! Tyra Banks Then & Now!
Tyra Banks Then & Now!